SNL 76/7 Passage-Independent Feeder Cells
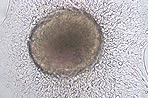
- Immortalized cell line derived from mouse fibroblast STO cell transformed with murine LIF and neomycin resistance genes
- Useful for culture of human or mouse iPS cells as well as ES cell growth
- Must be mitotically inactivated prior to addition of stem cells